Comprehensive Histology and Imaging Resources at UNE
The mission of the Histology and Imaging Core is to provide Center for Pain Research investigators and the greater UNE research community access to expertise, training, and specialized instrumentation related to tissue processing, sectioning, staining, immunohistochemistry, and microscopy.
The Core also offers services and training related to image analysis and image analysis software to guide investigators in choosing the best methods for presenting their data.
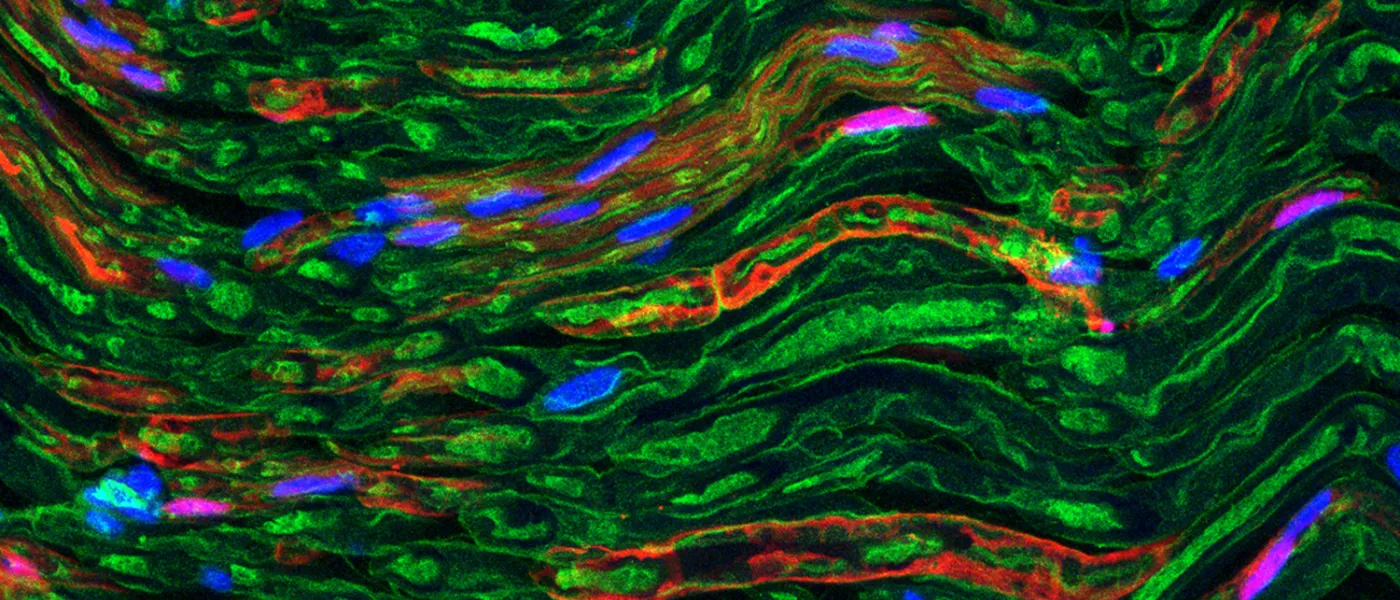
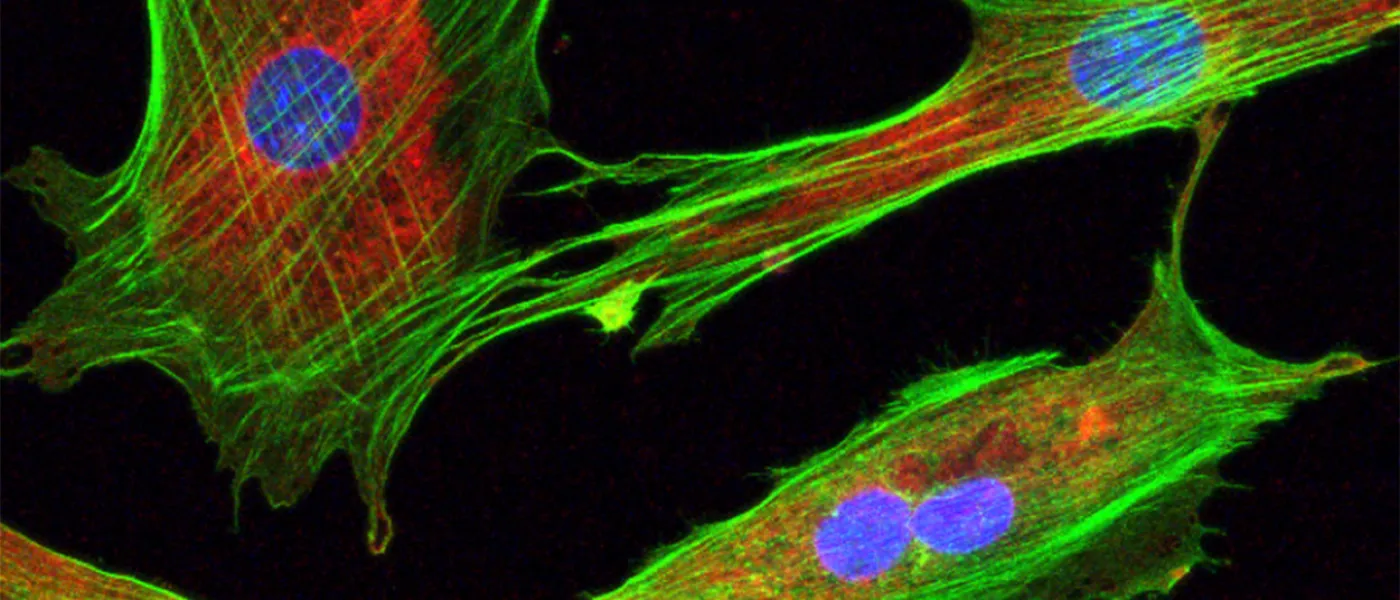
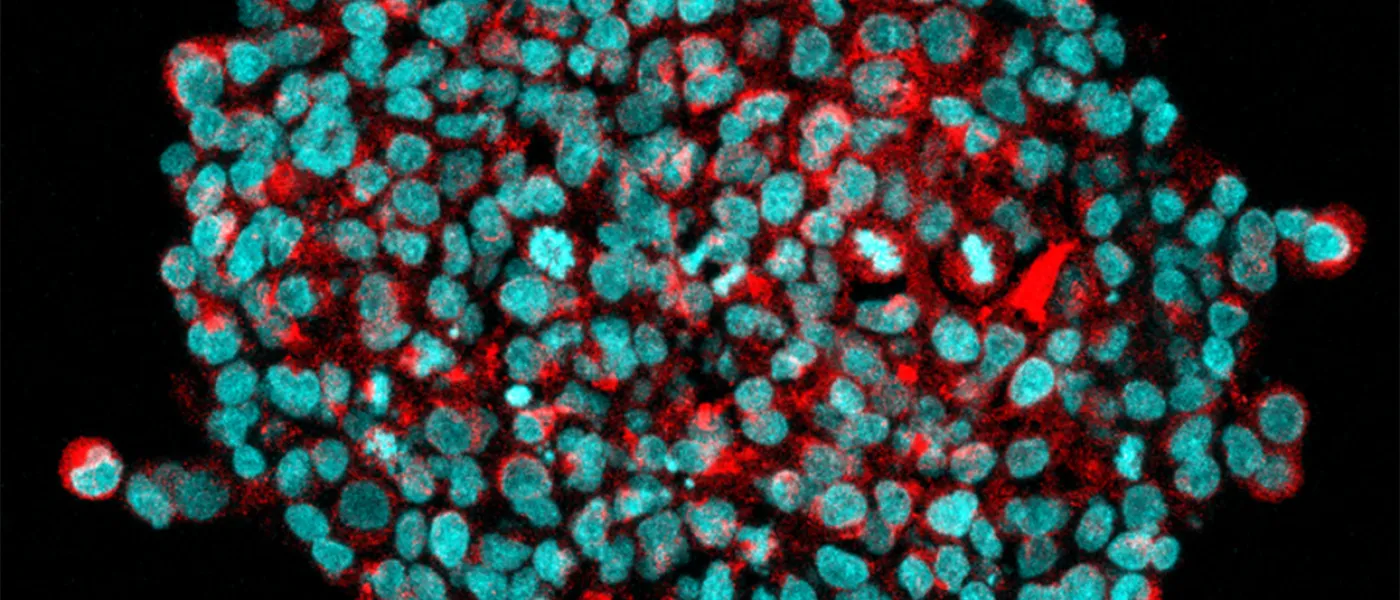
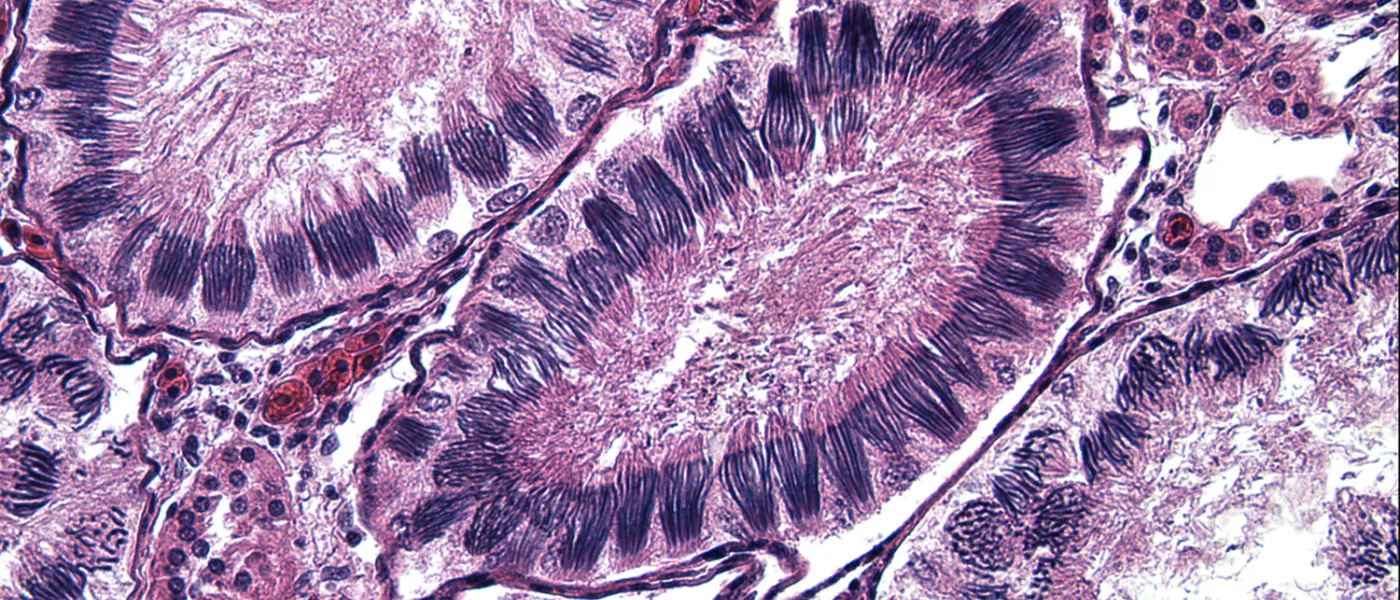
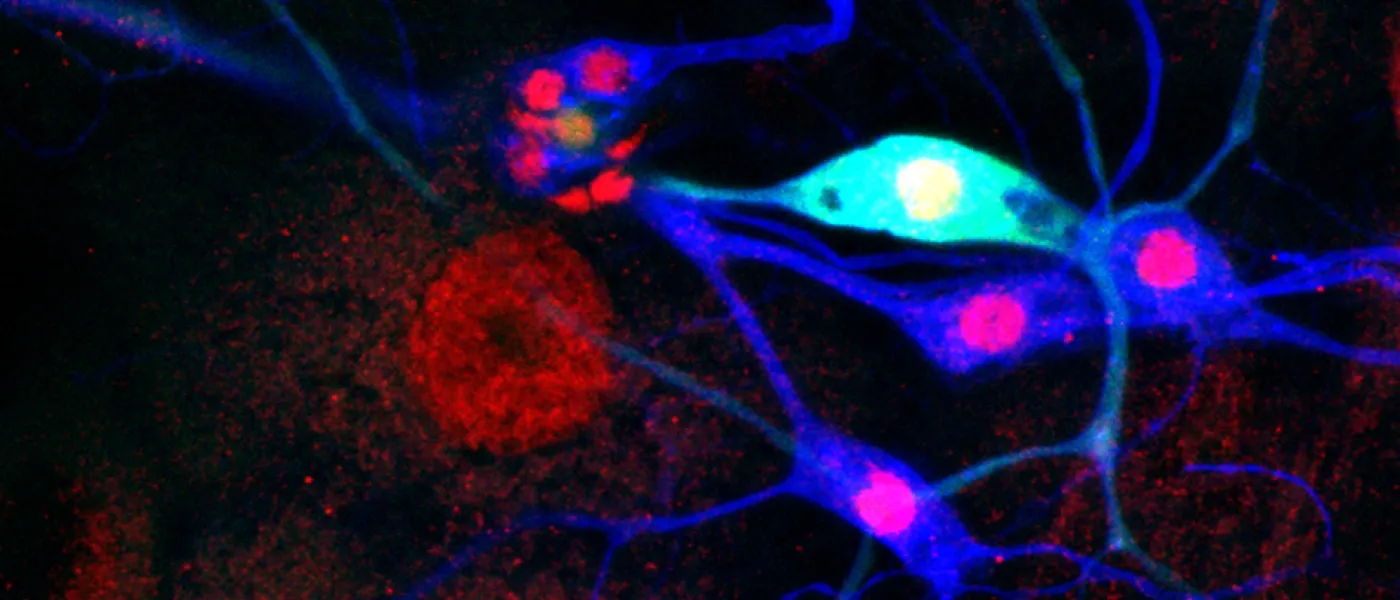
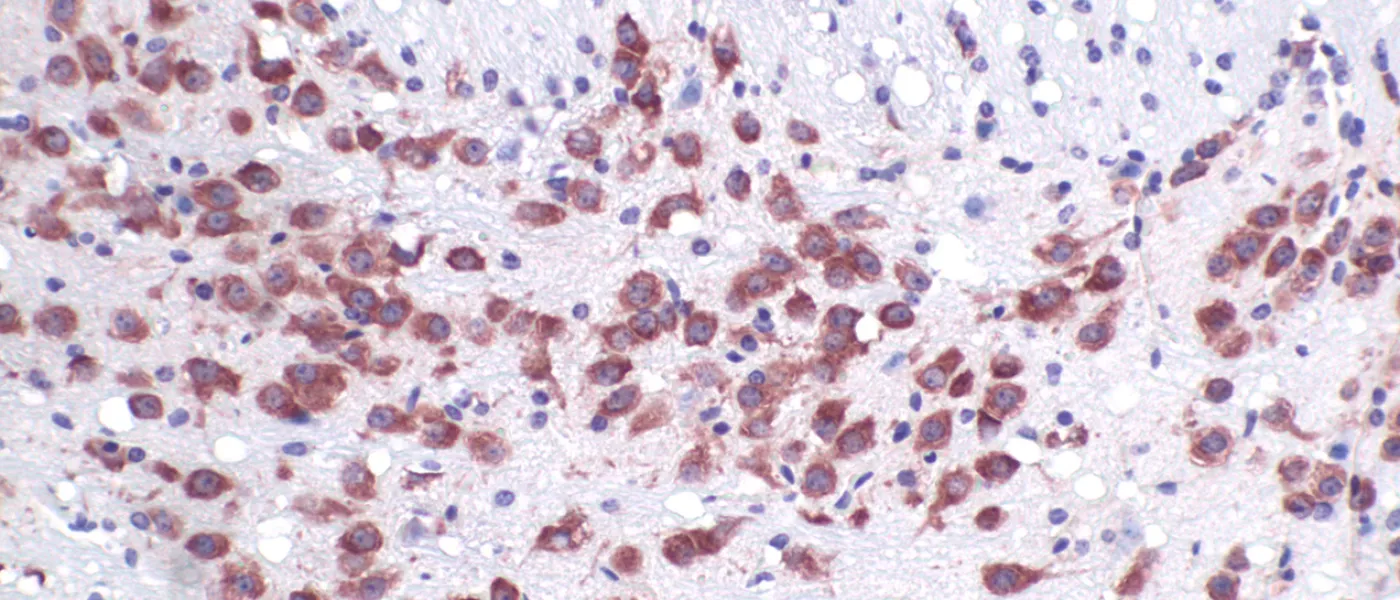
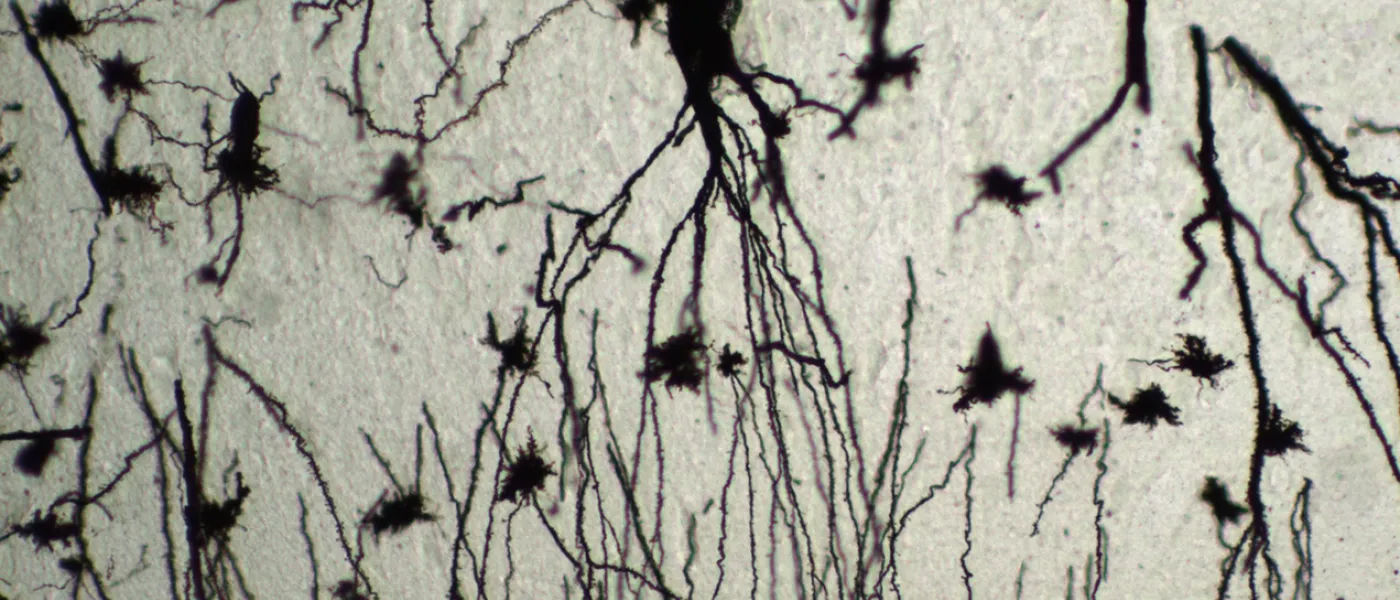
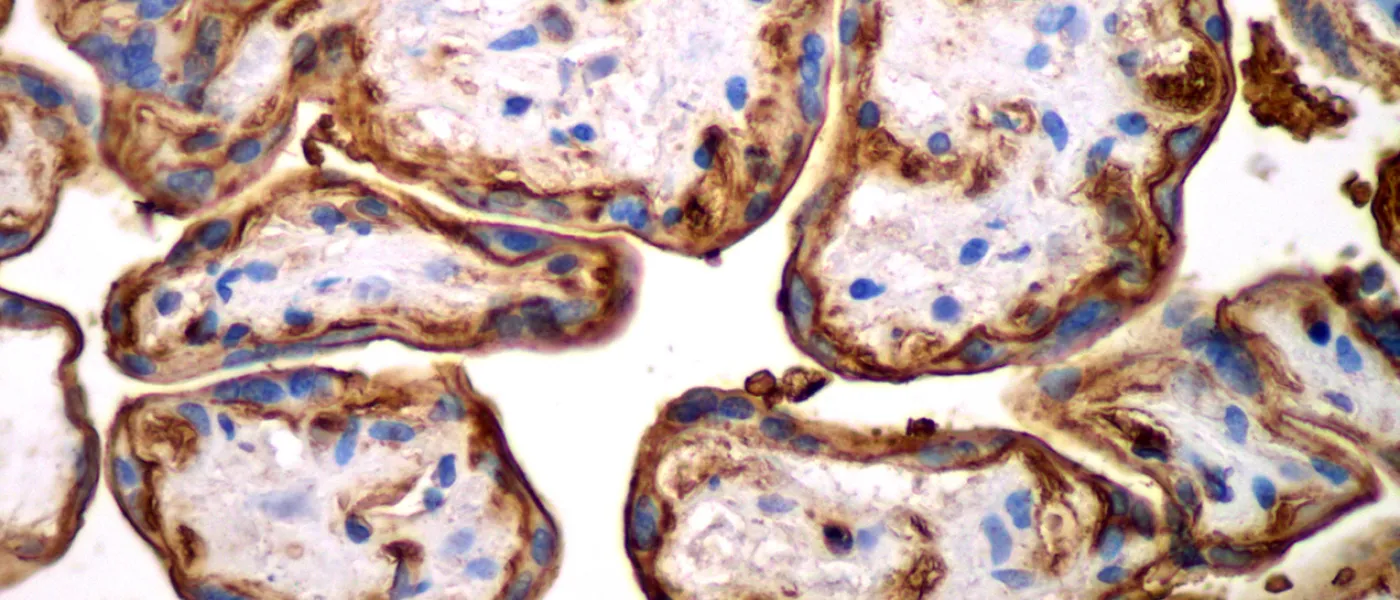
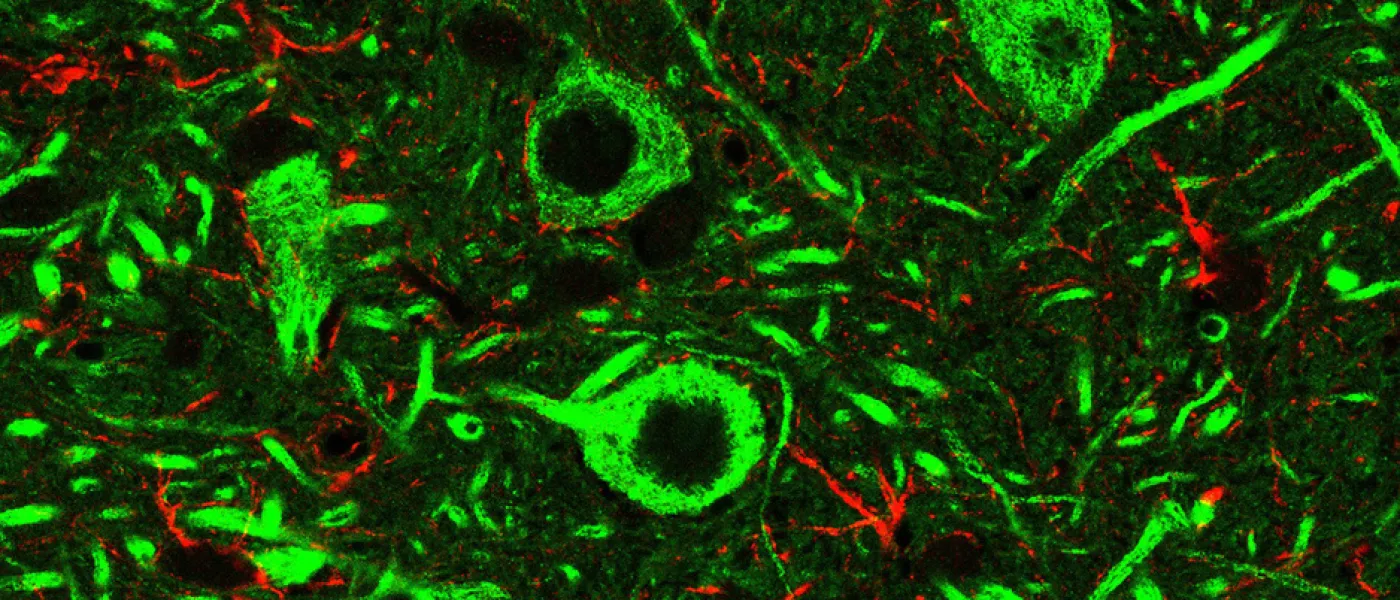
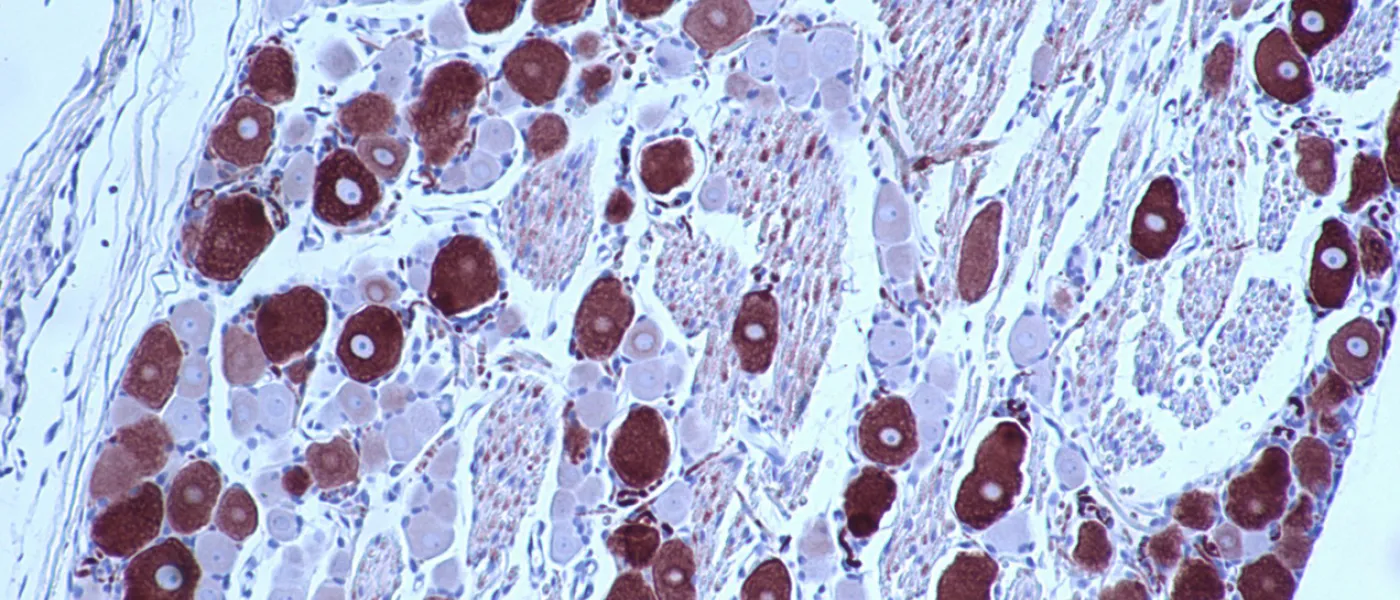
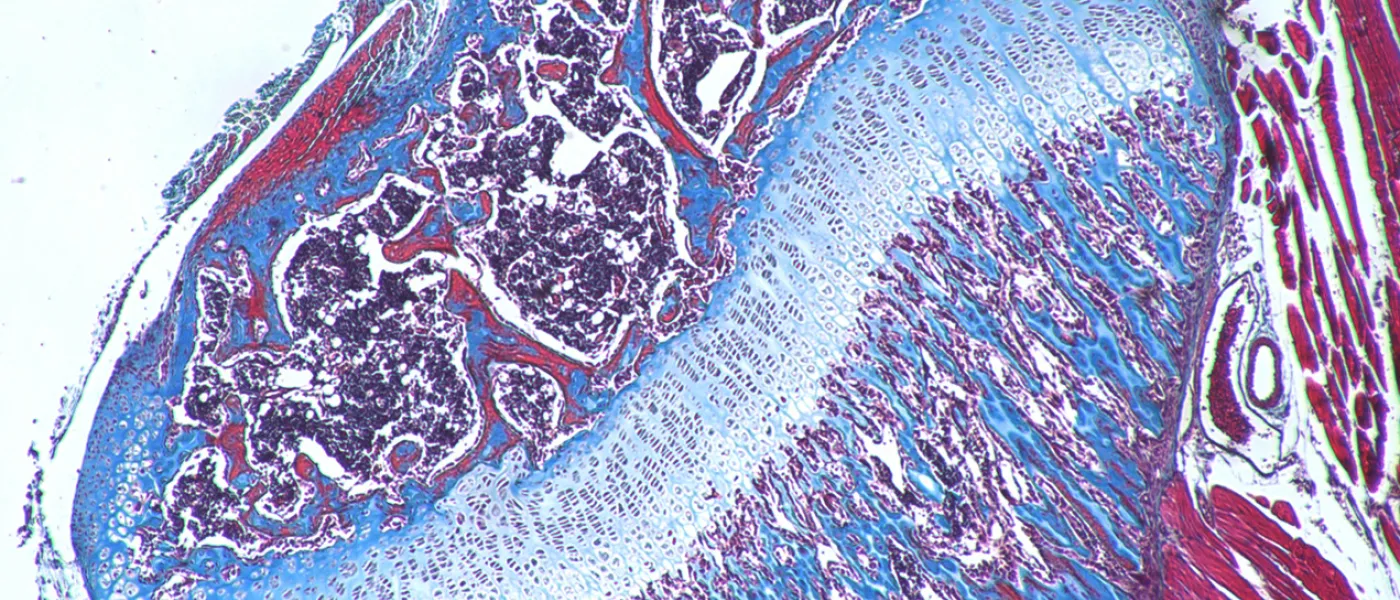
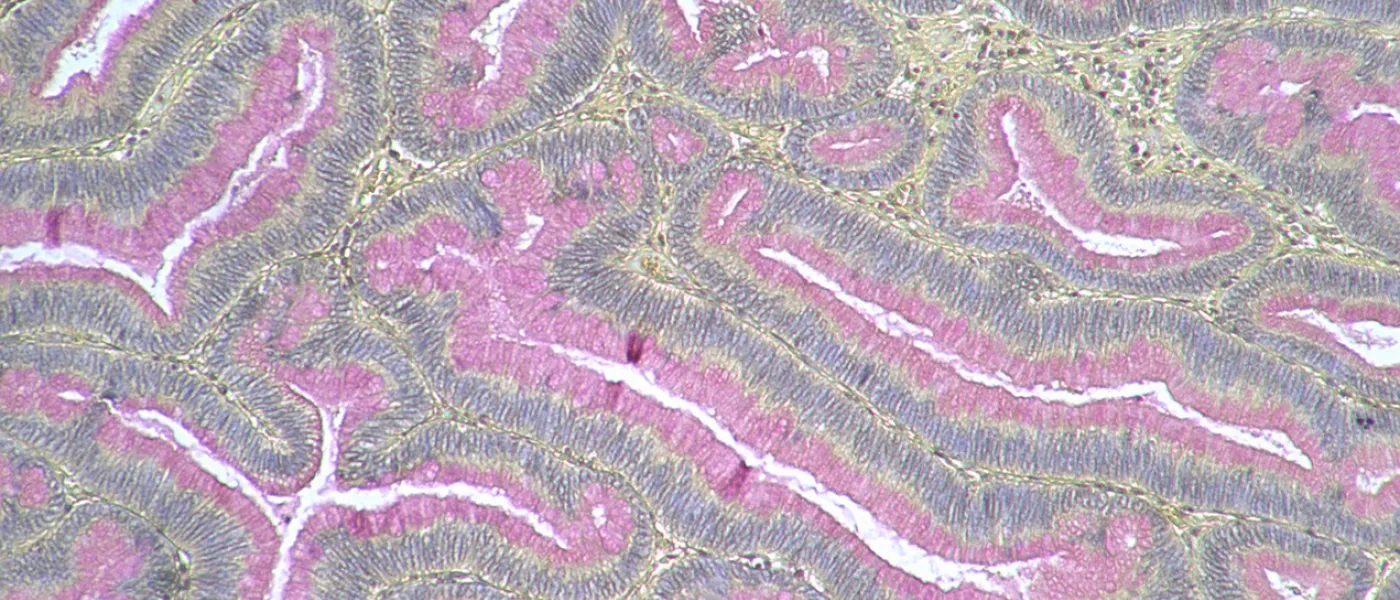
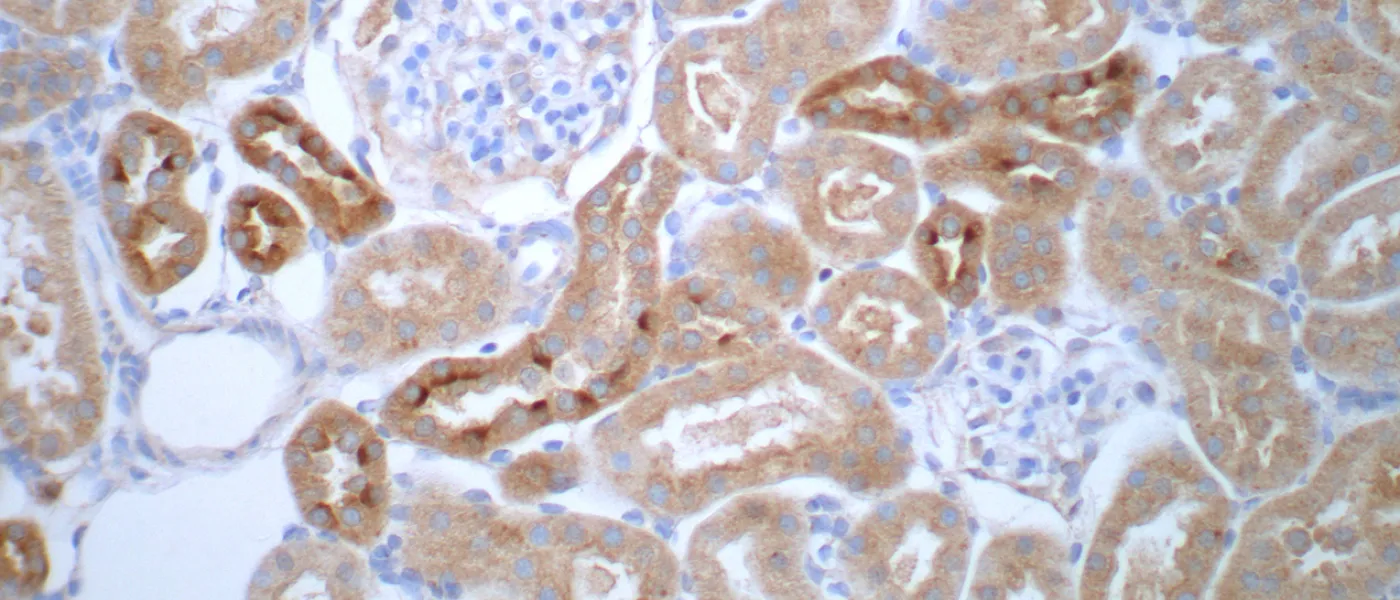
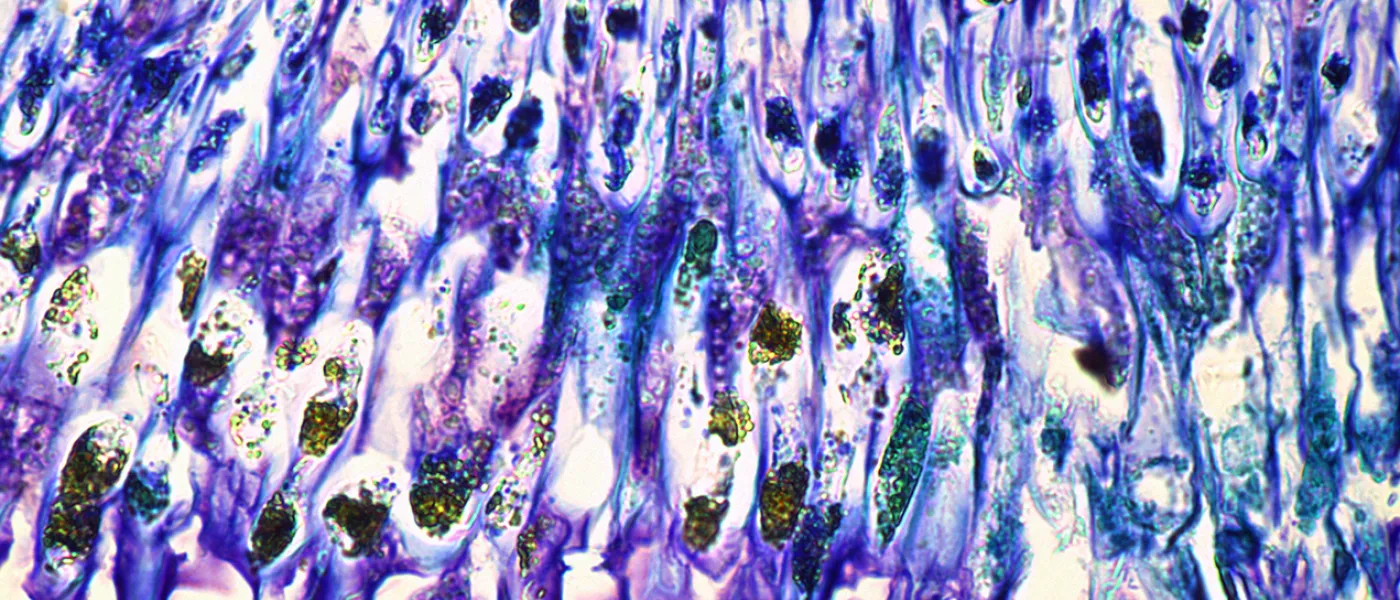
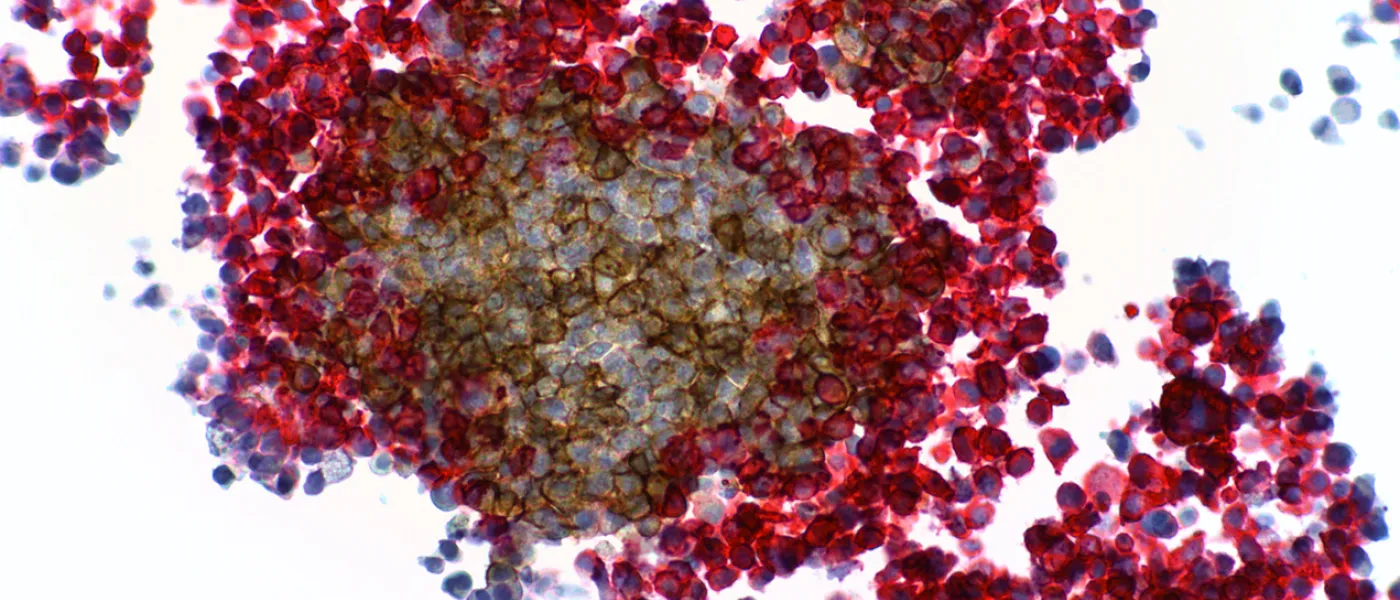
These images represent work completed by Histology and Imaging Core professional staff. All samples were processed on core equipment, and images captured on core microscopes. The gallery contains a wide variety of tissue types, including nervous tissue, bone, kelp, and drosophila larvae.
Services
The Histology and Imaging Core is fully equipped to perform a wide range of histological processing to suit investigators’ needs.
Service Fees
| Service | Unit | Internal Fee | External Academic Fee | External Commerical Fee |
|---|---|---|---|---|
| Miscellaneous labor, training | per hour | $41 | $63 | $126 |
| Decalcification, EDTA or formic acid | per sample, week | $3.62 | $4.92 | $9.85 |
| Paraffin processing, standard | per cassette | $2.24 | $2.89 | $5.78 |
| Paraffin processing, mega | per cassette | $2.80 | $3.45 | $6.91 |
| Embedding, Paraffin | per block | $2.18 | $3.20 | $6.41 |
| Embedding, Frozen | per block | $3.75 | $4.90 | $9.81 |
| Paraffin Microtomy, Routine | per slide | $1.89 | $2.76 | $5.52 |
| Paraffin Microtomy, Levels | per slide | $2.44 | $3.59 | $7.18 |
| Cryosectioning, up to 3 sections per slide | per slide | $2.36 | $3.13 | $6.26 |
| Cryosectioning, 3 or more sections per slide | per slide | $3.80 | $5.33 | $10.67 |
| H&E staining, manual | per slide | $2.44 | $3.26 | $6.51 |
| Special Staining, Tier 1 (TB, Picrosirius, PAS, Mucicarmine) | per slide | $3.13 | $4.42 | $8.84 |
| Special Staining, Tier 2 (Masson's Trichrome, Safranin O/Fast Green) | per slide | $4.65 | $6.53 | $13.06 |
| IHC/IF, Paraffin (user-supplied primary antibody) | per slide | $7.67 | $10.64 | $21.27 |
| IHC/I,F Frozen (user-supplied primary antibody) | per slide | $6.08 | $8.45 | $16.91 |
Sample Processing
The Histology and Imaging Core is fully integrated with the UNE's Behavior Core and can directly process samples collected by the Behavior Core.
Training
The Core also provides training to professional staff and students on equipment and techniques.
The core has cryostats, microtomes, and microscopes available for reservation. To avoid scheduling conflicts and maintenance issues, all equipment must be reserved using Outlook calendars, and individual users must be trained by core staff.
The Leica confocal has a separate training procedure consisting of two 2-hour supervised sessions, several informational videos, and an online quiz. The first training session is an introduction to the microscope guided by core staff. The second session entails imaging of a user-generated sample with minimal intervention from Core staff. Upon completion of a proficiency quiz, users can reserve time as desired.
For any training inquiries, please email hicore@une.edu.
Badges
The UNE Badging program is a non-credit bearing vehicle that digitally validates and documents knowledge, skill sets, qualifications, and competencies. Open to all learners, any participant who completes the learning activities and demonstrates achievement of the learning outcomes/competencies for a badge would be able to claim it. Sharing badges with graduate schools, potential employers, etc., is a great way to demonstrate some of the skills and competencies you have acquired that are not captured in an academic transcript.
For interested students, the core is now part of the UNE Badging program. Students will gain a knowledge-base and skillsets relevant to the core specialty.
Funding
Assistance is available for help to cover the fees for work performed by the research cores.





Publications
Karamchedu NP, Fleming BC, Proffen BL, Sant NJ, Portilla G, Parola LR, Molino J, Murray MM. Terminal sterilization influences the efficacy of an extracellular matrix-blood composite for treating posttraumatic osteoarthritis in the rat model. J Orthop Res. 2022 Mar;40(3):573-583.
Eaton VE, Pettit S, Elkinson A, Houseknecht KL, King TE, May M. Polymicrobial abscess following ovariectomy in a mouse. BMC Vet Res. 2019 Oct 24;15(1):364.
Sannajust S, Imbert I, Eaton V, Henderson T, Liaw L, May M, Barbe MF, King T. Females have greater susceptibility to develop ongoing pain and central sensitization in a rat model of temporomandibular joint pain. Pain. 2019 Sep;160(9):2036-2049.
Gjelsvik K, Follansbee T, Ganter G. Bone Morphogenetic Protein Glass Bottom Boat (BMP5/6/7/8) and its receptor Wishful Thinking (BMPRII) are required for injury-induced allodynia in Drosophila. Molecular Pain. Jan-Dec 2018;14:1744806918802703.
Sherman H, Gitschier HJ, and Rossi AE. A Novel Three-Dimensional Immune Oncology Model for High-Throughput Testing of Tumoricidal Activity. Front. Immunol., 23 April 2018.
Havelin J, Imbert I, Cormier J, Allen J, Porreca F, King T. Central Sensitization and Neuropathic Features of Ongoing Pain in a Rat Model of Advanced Osteoarthritis. J Pain. 2016 Mar;17(3):374-82.
Deal A, Erickson K, Shiers S, Burman M, Limbic system development underlies the emergence of classical fear conditioning during the 3rd and 4th weeks of life in the rat. Behavioral Neuroscience. Behav Neurosci. 2016 Apr;130(2):212-30.
Warner E, Krivitsky R, Cone K, Atherton P, Pitre T, Lanpher J, Giuvelis G, Bergquist I, King T, Bilsky E, and Stevenson G. Evaluation of a Postoperative Pain-Like State n Motivated Behavior in Rats: Effects of Plantar Incision on Progressive-Ratio Food-Maintained Responding. Drug Dev Research. Dec 2015; 76(8):432–441.
Burman M, Bilsky E. FAAH inhibitor OL-135 disrupts contextual, but not auditory, fear conditioning in rats. Behav Brain Res. 2016 Jul 15;308:1-5.
Burman MA, Simmons CA, Hughes M, Lei L. Developing and validating trace fear conditioning protocols in C57BL/6 mice. J Neurosci Methods. 2014 Jan 30;222:111-7.
Mclane V, Cao L, Willis C. Morphine increases hippocampal viral load and suppresses frontal lobe CCL5 expression in the LP-BM5 AIDS model. J Neuroimmunol. 2014 Apr 15;269(1-2):44-51.
Please remember to acknowledge the Center for Pain Research Histology and Imaging Core and the financial support provided by the NIGMS grant number P20GM103643 in manuscripts, grants, and presentations that include our work.
Contact
Location
University of New England Biddeford Campus
Pickus Center for Biomedical Research rooms 219/220
Histology and Imaging Core Email
Faculty and Professional Staff





