
What will you study? Laboratory Science Degree Curriculum Overview
B.S. in Laboratory Science Courses
The following are some examples of the exciting courses that you can take:
- Biochemistry
- Microbiology
- Physical Chemistry
- Advanced Lab
- Instrumental Analysis
- Parasitology
- Advanced Biochemistry Lab
- Quantitative Analysis
- Histology
- Human Anatomy, Physiology, and Pathophysiology
To see the full curriculum, view the academic catalog.
Honors Program
We offer qualified students the option of graduating with Honors. This includes significant scholarly, creative, or experiential activity under the direction of a faculty member. Interested students should consult with their advisor.

Career Paths for B.S. in Laboratory Science Majors
Whether you’re destined for a career in medical technology, quality control, biotechnology, or pharmacology, your major in Laboratory Science will provide the knowledge, laboratory skills, analytical abilities, and internship experience you need to succeed.
As a Laboratory Science graduate, there are many professions that you may explore, including:
- Biotechnology Researcher
- Biomedical Scientist
- Laboratory Technician
- Clinical Research Associate
- Research Scientist
- Teacher
- Science Journalist
- Toxicologist
- Forensic Scientist
- Phlebotomist
Career Advising for B.S. in Laboratory Science Students
Whether you have a specific career goal in mind or a vague idea of the field that interests you, Career Advising is here to help you plan your next step.
Laboratory Science Facilities
Our Laboratory Science program is housed in Peter and Cecile Morgane Hall, with additional teaching and research laboratories in the Alfond Center for Arts and Sciences (formerly the Alfond Center for Health Sciences) and biochemistry research laboratories in the Pickus Center for Biomedical Research. All of the facilities are located on our beautiful seaside campus in Biddeford.
Morgane Hall houses the general chemistry, general physics, and biochemistry teaching labs as well as some small research labs. The advanced chemistry teaching and research labs are located on the third floor of the Alfond Center and the first floor of the Pickus Center. Pickus has flexible biochemistry research space, where you work with faculty using a wide range of modern chemical and biochemical instrumentation in facilities designed to be conducive to learning and research productivity.
Experiential Learning in the Bachelor’s Degree in Laboratory Science
In our Laboratory Science program, you’ll engage in hands-on learning by working with faculty on research projects, conducting your own lab experiments, and getting on-the-job experience through internships – because at UNE we believe in learning by doing.
Internships for Laboratory Science Majors
You may complete an internship over the summer at sites including hospitals, private or government labs, educational institutions, or science centers. Or intern during the academic year with one of the prestigious employers with which we partner:
- IDEXX
- Lonza
- Jackson Laboratories
- Envirologix
- Katahdin Labs
For more information email the Academic and Career Advising Center at advising@une.edu.
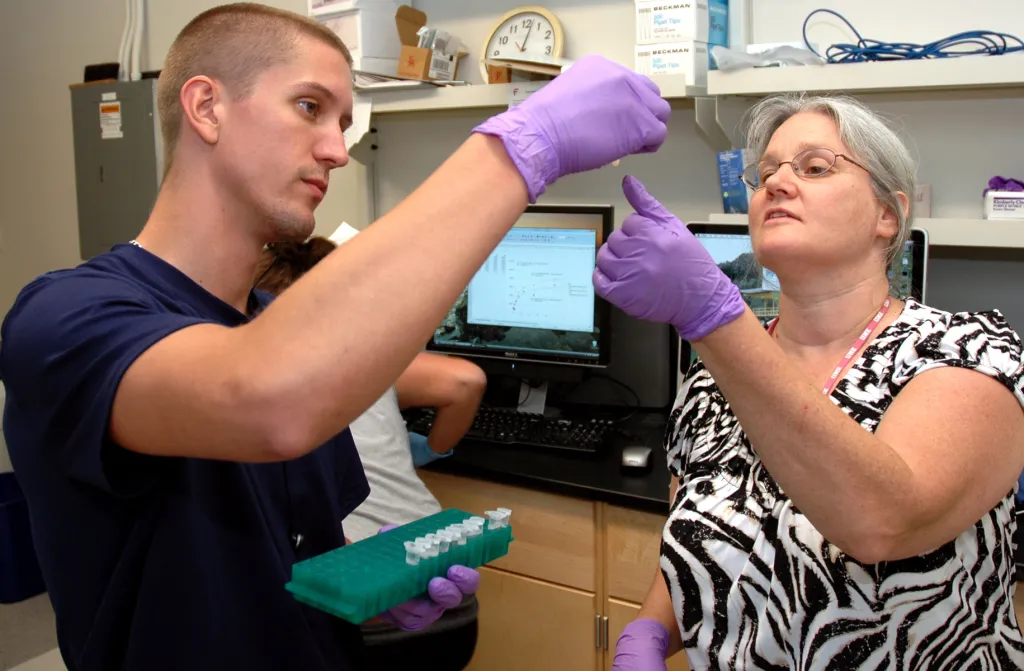
Research for Laboratory Science Students
We offer a breadth of research opportunities not commonly found at the undergraduate level – from synthesizing bioactive compounds and engineering protein materials to synthesizing small molecule drugs and exploring environmental toxicology,
- Collaborate with faculty on innovative research
- Engage in undergraduate research in your coursework
- Perform paid research
- Present at conferences and publish your findings
Amy M. Deveau, Organic and Medicinal Chemistry
There are three ongoing research projects in Amy Deveau's (Dr. D) lab:
- The design and synthesis of naltrexol derivatives for use as pain and addiction therapies
- The synthesis and biological characterization of tryptophan-based DNA intercalators
- The synthesis of medicinally active, nitrogen-containing compounds using green Suzuki Coupling methodology, and the extension of these experiments to the undergraduate organic lab curriculum.
Overall, Dr. D is passionate about finding ways to interest students in science and in learning new ways to integrate teaching and research in the classroom (i.e. chemical pedagogy).

Stephen Fox, Inorganic Chemistry
Along with interested UNE students and scholars, Dr. Fox plans to:
- Continue to characterize the physical and structural properties of the dicopper(I) model and its derivatives
- Further customize the naphthyridine with electron-donating groups to enhance the reactivity of the dicopper(I) center; selectively remove/replace the bridging groups to investigate the utility of the dicopper(I) center as a catalyst for reactions such as aziridination
- Investigate factors influencing the metal-metal separation through theoretical computational studies.

Amy Keirstead, Physical Organic Chemistry and Photochemistry
Research in Amy Keirstead's (Dr. K) group covers a wide variety of traditional areas, from synthetic organic chemistry to photochemistry, spectroscopy, materials science and green chemistry, and undergraduate students will gain experience in all of these areas. Two major pieces of equipment are used for this research: a photochemical reaction chamber, which is a steady-state instrument, and a nanosecond laser flash photolysis system, which uses a nanosecond laser and fast detection system to monitor reactions as they happen on the nanosecond to millisecond time scale.

Jerome Mullin, Analytical Chemistry
Several areas of research are being explored in Dr. Mullin's laboratory. One area is focused on the determination of heavy metal distributions in sediments from the Gulf of Maine, local rivers, and the Bering Sea, in an attempt to evaluate current levels of heavy metal pollution and identify degrees and sources of anthropogenic inputs of these elements. Of particular interest are the baseline heavy metal distributions in the Bering Sea, a relatively sparsely populated area of high primary productivity still considered to be largely unpolluted. Metals that have been determined to date, using atomic absorption spectroscopy and anodic stripping voltammetry, include cadmium, lead, and chromium.
Another area of research involves the spectroscopic and electrochemical characterization of a series of Group-14 and 15 metallacyclopentadienes (metalloles). These interesting compounds, which have unusual photoluminescence properties, show promise as monomeric units for the design of conducting polymers, for use in energy-transfer applications, as emitting species in light-emitting diodes (LEDs), and as components of chemical sensors. Luminescence quantum yields for the compounds recently have been determined and luminescence quenching studies are ongoing. Of special interest is the dramatic aggregation-induced emission (AIE) exhibited by many of these compounds, in which luminescence yields are increased by over two orders of magnitude compared to the unaggregated compounds. Current directions in this research also include the development of substituted metalloles designed for increased water solubility, improved luminescence yields, and for use as luminescent probes.
Photoluminescence is also the subject of research involving compounds containing a rare earth ion, e.g., Tb(III), Eu(III), or Dy(III) and the dicyanoaurate or dicyanoargentate ions. These compounds appear to have energy transfer characteristics that lead to "tunable" excitation of rare earth photoluminescence.
Other areas of interest include the development of fiber-optic probes based on fluorescence and/or chemiluminescence and the development of a chemiluminescence-based immunoassay system for the determination of trace amounts of dioxins.
Recent Grant Support: National Science Foundation, U.S. Fish and Wildlife Service, EOSAT Corporation, Pittsburgh Conference Memorial National Grants Program, American Chemical Society (ACS) Petroleum Research Fund (PRF), University of New England.

Deena Small, Biochemistry
One major research area in Dr. Small’s laboratory is the investigation of how proteins belonging to the Jagged1/Notch and Fibroblast Growth Factor (FGF) families interact and regulate adipocyte differentiation in response to hormones such as insulin. Since adipose tissue development and function is also dependent on the ability of the mature fat cells to secure a vascular system that will support its metabolic requirements, the study also includes analysis of Jagged1/Notch and FGF interactions as a regulator of angiogenesis- the formation of new blood vessels from the existing vasculature.
The second research project in The Small Laboratory is a toxicology study that characterizes the effects that Polybrominated Diphenyl Ethers (PBDEs), chemicals used as flame retardants in a variety of household goods, have on mesenchymal stem cell differentiation. In particular, research is focused on analyzing the effects that these endocrine-disrupting chemicals have on mesenchymal stem cell growth dynamics and their ability to differentiate into adipocytes and osteoblasts. These studies also document the impact that early PBDE exposure has on the formation of adipose and bone. The ultimate goal of both projects is to understand the connection that these signaling mediators have on human health and disease. Both research projects use a combination of cell culture and in vivo biomedical models such as the mouse and zebrafish. Techniques include standard biochemical/molecular biology methods including quantitative RT-PCR, immunoblot, and ELISA. In addition, Dr. Small employs molecular techniques such as cloning to produce transgenic mice and zebrafish for the studies. These projects involve collaborations with scientists from UNE, Maine Medical Center Research Institute, SUNY Geneseo, University of Maine, Bates College and University of Quebec, Montreal.

John Stubbs, Physical Chemistry
The first area is focused on DNA sensor microarray technology, which is composed of single-stranded DNA oligomers bound to a surface. The development of this technology relies on knowledge of DNA denaturation or "melting" and hybridization transitions, and their sensitivity to many variables has left several questions unanswered, such as the mechanism by which changes in physical environment between solution and bound DNA act to influence hybridization, or competitive adsorption of nearly identical sequences.
The second area attempts to improve upon separation technology using fairly innocuous materials such as supercritical carbon dioxide and polyethylene glycol to achieve what traditional processes do with more detrimental materials. Molecular simulations allow the modeling of such systems with the goal of optimizing solvation conditions and is done by carrying out calculations with varying thermodynamic (e.g. temperature and/or pressure) conditions and compositions.

James Vesenka, Physics
"Dr. V” conducts research in the self-assembly process of four-stranded “G-wire” DNA using scanning probe microscopy (SPM). He is interested in understanding the kinetics of the self-assembly process and the interaction of the G-wire DNA with substrates and double-stranded DNA. G-wires make exciting candidates for possible nano-electronic devices, so he is interested in their electronic properties and the ability to manipulate the self-assembled structures at the microscopic level with the SPM. With the advent of fast local area networks he has safely connected the sensitive SPM to the noisy environment of the introductory physics lab using virtual network computing.
Dr. V also conducts research in the area of physics pedagogy. He is developing more effective means for teaching introductory physics through the use of multiple representation learning tools, also called “modeling,” which includes better student evaluation procedures. He has organized two workshops to train pre-service and in-service science teachers in Maine and California for the summer of 2000.
Grant support: Research Corporation, National Science Foundation, Maine Mathematics and Science Alliance.

Apply Today
Ready to begin your future in UNE’s B.S. in Laboratory Science degree program? Get started today.
Laboratory Science FAQ
What is a laboratory science major?
A major in laboratory science is all about preparing students for work in a variety of modern laboratory settings, ranging from biotechnology labs to quality control labs to academic, medical, or industrial research labs.
UNE offers a Bachelor of Science (B.S.) degree program in Laboratory Science, preparing graduates for careers in modern laboratory settings, including biotechnology, quality control, research, and academic or medical labs. The major focuses on classic and state-of-the-art lab techniques and instrumental methodology, providing a strong foundation in chemistry, biology, and biochemistry.
Learn more about the Bachelor of Science (B.S.) Degree in Laboratory Science at UNE
What can you do with a laboratory science degree?
Whether you’re destined for a career in medical technology, quality control, biotechnology, or pharmacology, majoring in Laboratory Science will provide the knowledge, laboratory skills, analytical abilities, and internship experience you need to thrive.
Graduates of UNE's B.S. in Laboratory Sciences undergraduate degree program have embarked on diverse career paths holding positions such as biotechnology researchers, biomedical scientists, laboratory technicians, clinical research associates, research scientists, teachers, science journalists, toxicologists, forensic scientists, and phlebotomists.
UNE students are encouraged to utilize the resources available at our Academic and Career Advising Center. It doesn’t matter if you have a clear career goal or just a general interest in a field.
What are the courses under a laboratory science degree?
A laboratory science degree encompasses a wide range of laboratory courses in chemistry, biochemistry, and biology. The goal is to furnish graduates with a comprehensive set of relevant laboratory skills that can be effectively applied across various sectors, including chemical, biological, and medical industries.
The following are some examples of the exciting courses that you can take as a Laboratory Science major at UNE:
- Biochemistry
- Microbiology
- Physical Chemistry
- Advanced Lab
- Instrumental Analysis
- Parasitology
- Advanced Biochemistry Lab
- Quantitative Analysis
- Histology
- Human Anatomy, Physiology, and Pathophysiology
How many years is a laboratory science degree?
The time it takes to complete a laboratory science degree varies depending on the specific degree you're aiming for. For instance, if you're considering UNE’s B.S. in Laboratory Science, it typically entails around 120 credit hours, which translates to about four years of full-time study. This includes:
- 42-46 credits of College of Arts and Sciences (CAS) core curriculum
- 64 minimum program credits of program-required courses
- 12–14 credits of flexible program-required courses
- Open electives as needed to reach 120 credits
Which universities offer the best laboratory science degree programs for me?
When you're trying to figure out which college or university offers the best laboratory sciences degree program for you, there are a bunch of things you need to think about.
Faculty
When choosing a laboratory science program, evaluate the qualifications and research interests of the faculty. Ensure that their expertise and research areas match your interest in laboratory science and are in line with your academic and career goals.
One of the standout features of UNE’s undergraduate laboratory science major is the opportunity to collaborate closely with a faculty member on a captivating research project. Below, you'll discover detailed descriptions of the research projects currently conducted by our faculty members:
- Amy M. Deveau, Ph.D. - Organic and Medicinal Chemistry
- Stephen Fox, Ph.D. - Inorganic Chemistry
- Amy E. Keirstead, Ph.D. - Physical Organic Chemistry and Photochemistry
- Jerome Mullin, Ph.D. - Analytical Chemistry
- Deena J. Small, Ph.D. - Biochemistry
- John M. Stubbs, Ph.D. - Physical Chemistry
- James Vesenka, Ph.D. - Physics
Meet UNE faculty in the School of Mathematics and Physical Science
Curriculum
When contemplating a laboratory science program as a prospective college student, evaluate the curriculum's alignment with your interests. Investigate areas such as general laboratory science, organic laboratory science, physical laboratory science, or related subjects, and inquire about research opportunities and internships within the field of laboratory science.
UNE's Laboratory Science curriculum focuses on both traditional and advanced laboratory techniques, providing a strong foundation in chemistry, biology, and biochemistry. Through a diverse range of lab courses, you'll acquire a versatile set of skills applicable in modern laboratory environments, including academic, medical, and industrial labs.
We also believe in experiential learning (learning by doing). In our Laboratory Science program in Maine, you’ll engage in hands-on learning by working with faculty on research projects, conducting your own lab experiments, and getting on-the-job experience through internships.
Learn more about UNE’s Laboratory Science curriculum
Career Objectives
Select a laboratory science degree program that aligns with your career aspirations and goals. Explore the available resources to assist you in planning your future path.
With a thoughtful selection of elective courses, graduates aspiring to become medical technologists can position themselves favorably for acceptance into accredited hospital internship programs in medical technology. Notably, UNE's recent laboratory science graduates have achieved success in diverse career opportunities across industries and have excelled in pursuing advanced studies in graduate programs.
UNE Laboratory Science students may complete an internship over the summer at sites including hospitals, private or government labs, educational institutions, or science centers. You could also intern during the academic year with one of the prestigious employers with which we partner:
- IDEXX
- Lonza
- Jackson Laboratories
- Envirologix
- Katahdin Labs
UNE students can work with our Academic and Career Advising Center. It doesn’t matter if you have a clear career goal or just a general interest in a field.
Learn more about Career Advising at UNE
Learning Environment
Decide whether you want to stay within your state or explore opportunities in another state. Evaluate whether the student and faculty community aligns with your desire for inspiration and intellectual stimulation.
Ranked a “High Research Activity” university by the Carnegie Classification, UNE in Maine boasts all the advantages and resources that you would expect at a large institution. Yet we provide a small, intimate college environment that gives you one-on-one attention and access to sophisticated equipment that are uncommon at the undergraduate level.
Read testimonials from students in the School of Mathematical and Physical Sciences
UNE’s Laboratory Sciences program is situated in Morgane Hall, with additional labs in the Alfond Center for Health Sciences and the Pickus Center for Biomedical Research, all on our scenic seaside campus in Biddeford, Maine - one of three unique campuses that provide diverse learning opportunities.
Morgane Hall houses the general chemistry, general physics, and biochemistry teaching labs as well as some small research labs. The advanced chemistry teaching and research labs are located on the third floor of the Alfond Center and the first floor of the Pickus Center. Pickus has a flexible biochemistry research space, where you work with faculty using a wide range of modern chemical and biochemical instrumentation in facilities designed to be conducive to learning and research productivity.
Learn more about UNE’s campuses and how you can visit us
Financial aid
Does the school you are considering offer financial aid or scholarships to help you in your education?
A UNE education is very affordable. Our tuition is 16% below the average tuition for private universities in New England. All incoming full-time undergraduate students at UNE will receive Merit Scholarships in amounts from $5,000 to $22,000 per year.
Learn more about grants and scholarships available to students at UNE
UNE's Student Financial Services includes both Financial Aid and Student Accounts offices. Our program maximizes available institutional and federal funds. To discover ways to fund your education, reach out at sfs@une.edu or (207) 602-2342 during our Monday–Friday 9 a.m.–3 p.m. office hours.

